Quality
Acti-Med - fit for quality
One of Acti-Med's main goals is to provide its customers with products that are of the highest quality possible.
We ensure the quality of our products by means of comprehensive incoming goods inspections, constant in-process controls (during the entire period of production) and outgoing goods inspections of the finished products intended for our customers.
In consultation with the customer, test steps that are necessary to ensure a high-quality production plan are determined. While using the test plan appropriate measurements are made during production.
Quality control
In order to make sure quality is to standard at every step of the process, extensive quality controls are carried out before the start of production.
- Ensuring quality through comprehensive incoming goods inspections of the supplier parts according to agreed upon specifications
- Sample test plans (DIN 2859, C = 0 error strategy)
- Constant in-process controls that monitor quality during production.
- Statistical analysis on process position determination.
- Outgoing goods inspection of finished, and customer-specific, products using a CAQ system.
- Close-meshed monitoring system that continuously checks the production processes, especially the cleaning.
- Final batch-based evaluation, documentation and approval of a production order.
Quality assurance
- Clearly defined processes make it possible to reproduce the production methods.
- Continuous training of our employees ensures the guarantee of quality requirements.
- Use of modern (automated) measuring systems
- Implementation of these tasks by current and extensive computer systems such as CAQ (Babtec), CAD (Solid Works), PDM / PLM (CIM-Database) or ERP (Bäurer B7).
Quality Planning
All of our activities are oriented towards the customer and the specific product requirements.
In order to ensure the safe production and the requirements of the products, Acti-Med is subject to, and certified under the quality management system according to DIN EN ISO 13485:2016, and we are subject to the annual surveillance audits of our Notified Body (certifier).
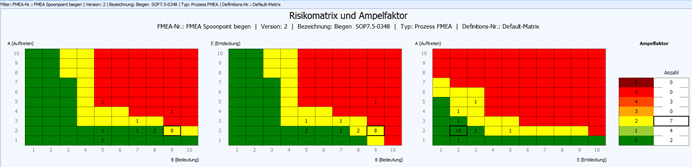
We respect the principles of ISO 15378 and GXP (GMP, GDP, etc.) and apply them product-related. Risk analysis (FMEA based) belongs to our graphic GMP.
It is important to us that we are able to test everything we produce. Whether you opt for production in our factory in Germany or our subsidiary Acti-Med Polska (Poland), all our products are subject to strict quality requirements!
To ensure this, regular audits and batch-related incoming goods inspections take place, which can always be tailored to the wishes of the customer.
Biological investigations
he cannula cleaning process at Acti-Med is validated for low endotoxin levels
As part of a monitoring plan, bioburden and endotoxin values of the cannulas and the final cleaning basin and water treatment plant are also regularly checked.
With these measures, you can be sure that the needles manufactured by Acti-Med are fully in line with your stringent biological purity requirements.
Validation and Documentation
Upon request, we can provide you with qualification and validation documentation tailored to your requirements. Our existing plants and main processes are qualified / validated. The results are available to both new and existing customers at any time.
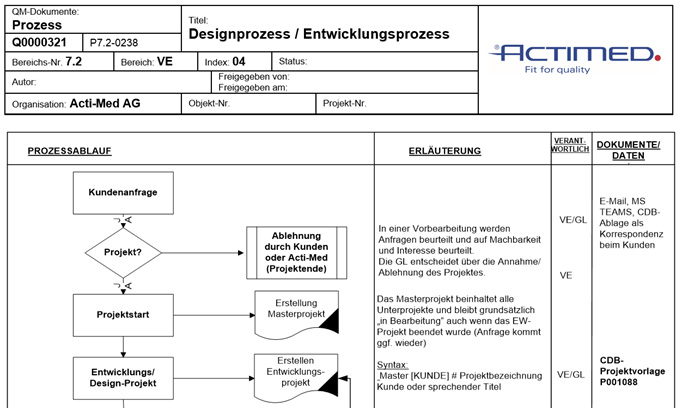
We document our development processes by means of dedicated processes and checklists, which are processed during the project phase. Through regular consultation you will be kept informed of the progress of the project, and all documents will be secured within the given framework.
Documentation about the project management system CIM-Database
Through our Product Lifecycle Management (PLM) system CIM DATABASE we are able to document tasks, documents, know-how and project progress and to share them with our customers. From the start of the project to the delivery of the first series and beyond, it is possible to store all activities centrally in one place and also to find them again when needed. Last but not least, the status control of the articles and drawings enables us to precisely control projects and orders.
All product-related documents (from the raw materials data sheet to the COC of the finished product) are always linked to the part number. Upon completion of the project, Acti-Med staff will have permanent access to documents generated during the development phase, such as product specifications, processes and product drawings.
The database is distributed over several servers. Archiving takes place according to defined plans so that at least the legal and the archiving periods required by the standard are guaranteed.
QM-dokuments in CIM-Database
In our PDM system, we can retrieve all documents specifically for an article. For example, SOPs or other QM-relevant documents are available. Processes, instructions, and delivery specifications follow all defined workflows for release and archiving.